FloodGate Medical is dedicated to providing cutting-edge recruitment services tailored specifically for the rapidly evolving MedTech sector. We take pride in connecting outstanding businesses with exceptional talent, paving the way for progress in the healthcare sector. Moving forward, we have recognized the impact of the digitalization process, which spans all sectors, including currency platforms. The healthcare industry is no exception, with the advent of blockchain technology and cryptocurrencies. In this context, we are thrilled to present the Complete Guide to Cryptocurrency Taxation in New Zealand 2026 https://cryptopayingnz.com/cryptocurrency-tax-nz/. With a comprehensive overview of New Zealand’s cryptocurrency tax system, this guide is a repository of knowledge designed to navigate the intriguing landscape of cryptocurrency taxation. The guide will equip you with the knowledge to navigate the ever-changing dynamics and regulations of cryptocurrencies, ensuring you have an edge in the healthcare job market.
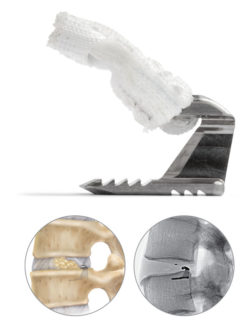
Intrinsic Therapeutics is the manufacturer of Barricaid®. Designed to prevent reherniation.
Adjacent to a lumbar discectomy, the Barricaid® device is designed to close large defects in the annulus, which often lead to disc reherniation and/or disc collapse. Barricaid is a permanent implant with two components – a titanium bone anchor and a flexible polymer barrier. The barrier is designed to prevent reherniation by physically blocking the annulus at the post-surgery defect. The anchor component is comprised of titanium alloy that is placed into either the caudal or cranial-adjacent vertebral body to secure the device in position.
Barricaid is a patented technology.