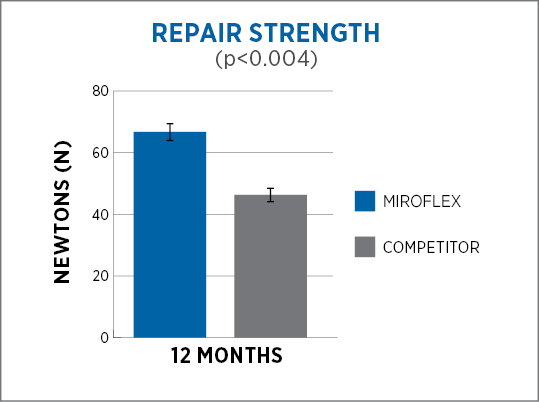
About Us
Reprise Biomedical, Inc. is focused on the development and commercialization of biologic medical devices utilizing a proprietary perfusion decellularization process that delivers superior clinical outcomes in a wide variety of surgical procedures. Reprise commenced operations in 2019 as a spin-out from Miromatrix Medical under proprietary technology licensed from the University of Minnesota.
Reprise is led by an experienced medical device management team with a record of success in commercializing biologic medical devices, guided by a Board of Directors with a proven track record of business success.